November 24 2022
CO2 and pH
Carbon is one of the most important factors for microalgae growth because it is a key ingredient in photosynthesis.

Microalgae take up CO2 from the seawater around them and then use light energy to convert CO2 to organic molecules like carbohydrates. So it is important that algae are provided with lots of carbon so that their growth and photosynthesis aren’t limited!
However, this gets a bit tricky when we consider the second topic of this article: pH. When CO2 dissolves in water it dissociates to form HCO3– and H+. The H+ produced makes the water more acidic and reduces the pH.
This chemistry may be familiar in the context of ocean acidification. Extra CO2 in the atmosphere from human activities is dissolving in the ocean which reduces the pH. For most of the 20th century seawater pH was normally around 8.2, but many locations are seeing levels below 8 regularly now.
When microalgae grow the opposite of acidification happens: they take up CO2 from the surrounding water and the pH increases. This happens in both fresh and salt water. The pH of fresh water is more variable than sea water, but generally it is more acidic with a range of 6 – 8 (Fondriest Environmental 2013). Deviating from the natural pH ranges too much can reduce microalgae growth or even crash the culture.
Carbon and pH control are both important for microalgae culture so that 1) carbon doesn’t limit photosynthesis and 2) pH is in an optimal range for growth. Because they are so interrelated, it makes sense to control them together. Some options for pH control and carbon addition are:
- Swirling or bubbling with air – this increases the amount of gas exchange between the culture and the air which will add more CO2 (because air contains 0.04% CO2, Engineering Toolbox). However, this works better with smaller, more dilute cultures (e.g. flasks, carboys).
- CO2 bleed into airflow – this adds a continuous amount of CO2 which supplements the CO2 in the air. In combination with pH monitoring this can work well; however, it is important to consider changes in CO2 demand if you are using this approach.
- CO2 additions based on pH feedback – determine an appropriate pH setpoint and add CO2 as required to maintain this setpoint as algae grows. This is more complicated than the previous two strategies but is the best for maintaining sufficient CO2 and stable pH for algae growth. An example of what this looks like in the PBR 1250L is below (Fig 1).
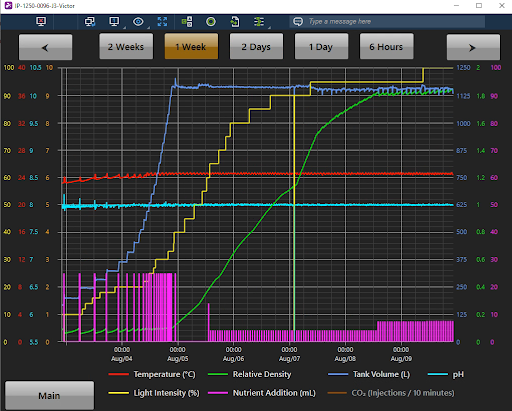
For more information on pH and carbonate chemistry in water check out this article by Fondriest Environmental:
